FFE separation of cell lysates
Complex protein mixtures of cell lysates can be separated in native or denaturing conditions.
Our standard workflow for cell lystaes is a two dimensional separation with denaturing IEF-FFE in the first step and SDS-PAGE on a second step.
Subsequnetly, the bands can stained with CBB and cut out of the gel for further mass spectrometry analysis and identification.
Alternatively the fractions can also be analyzed by HPLC-MS instead of SDS-PAGE.
Depending on the needs for the separation, the width of the separation area can also be adjusted for high throughput or high resolution, enabling separations down to only 1 Minute dwell time.
Comparison of IEF-FFE and iZE-FFE separation on native HeLa cell lysate.

IEF separation
HeLa cell protein lysate was separated under native conditions on IEF mode, pH gradient pH 3 – pH 10 with ampholyte, glycerol and polymere (HPMC). Second dimension SDS-PAGE, silver stained.
M=marker proteins
S=unseparated sample
#=FFE fraction
iZE separation
HeLa cell protein lysate was separated under native conditions on iZE mode, no ampholytes, no glycerol and no polymere (HPMC) were added. Second dimension SDS-PAGE, silver stained.
M=marker proteins
S=unseparated sample
#=FFE fraction
Whereas protein interaction and precipitation occured at the acidic area of the IEF separation, the iZE mode kept the solubility of the sample unchanged and thus a high resolution separation could be established along the whole width of the chamber.
Also a higher number of high molecular weight proteins >100kDa could be detected in iZE mode, compared to the IEF separation.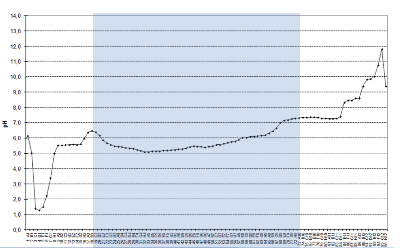
The graph on the right shows the iZE-pH profile of the cell lysate separation. The fractions 19-73 (blue marked area) were applied to SDS-PAGE.
(Added on July 26th 2013)
Example: cell lysate denaturing separation, one segment:

Separation of cell lysate of thyroid carcinoma cells.
As first dimension a pH 4 to pH 9 IEF-FFE gradient was used on 1/7 of the separation area.
The separation time of the sample through the chamber is 64 seconds.
After collection, the sample was applied to SDS-PAGE and silver stained.
M=marker proteins
S=unseparated sample
#=FFE fraction
This technique enables real high throughput fractionation for decreasing the complexity of samples.
Example: cell lysate denaturing separation, two segments:
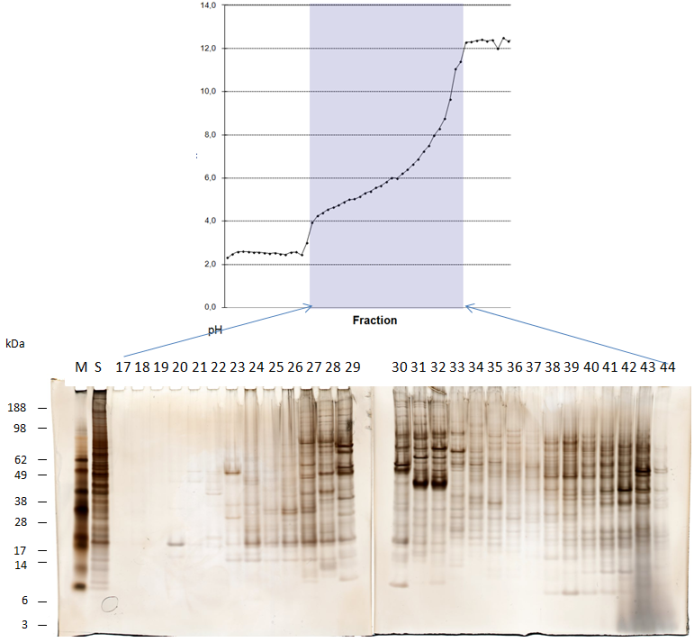
Separation of cell lysate of thyroid carcinoma cells.
As first dimension a pH 3 to pH 10 IEF-FFE gradient was used on 2/7 of the separation area.
The separation time of the sample through the chamber is 2 Minutes.
After collection, the sample was applied to SDS-PAGE and silver stained.
M=marker proteins
S=unseparated sample
#=FFE fraction
This technique combines reasonable throughut for separation with higher resolution.
Example: cell lysate denaturing separation, three segments, no polymer:
Separation of cell lysate of thyroid carcinoma cells.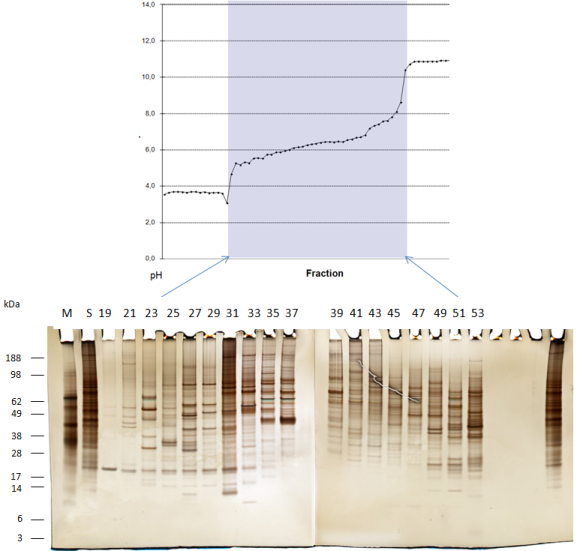
As first dimension a pH 3 to pH 10 IEF-FFE gradient was used on 3/7 of the separation area. The buffers here contain no polymer like HPMC.
The separation time of the sample through the chamber is 3.5 Minutes.
After collection, the sample was applied to SDS-PAGE and silver stained.
M=marker proteins
S=unseparated sample
#=FFE fraction
This technique enables unlimited possibility for concentration after FFE.